ALTERNARIA BLACKSPOT PHYTOTOXINS: NEW STRATEGIES FOR DETERMINING SPECIFIC DISEASE RESISTANCE TRAITS
M. S. C. Pedras, I. L. Zaharia, K. C. Smith, Y. Gai, and D. E. Ward
Department of Chemistry, University of Saskatchewan, 110 Science Place, Saskatoon SK S7N 5C9, Canada
ABSTRACT
Alternaria blackspot is one of the most destructive diseases affecting brassica crops worldwide, including the economically important oilseeds rapeseed/canola (Brassica napus and B. rapa). Destruxin B is the major host-selective phytotoxin produced by the causative agent of Alternaria blackspot, the blackspot fungus (Alternaria brassicae (Berk.) Sacc.). As part of a research program aimed at understanding mechanisms of plant disease resistance, we are evaluating the role of host-selective phytotoxins. Because white mustard (Sinapis alba) is resistant to Alternaria blackspot, we were interested in examining its response to the blackspot phytotoxins. Thus, the transformation of destruxin B by white mustard, canola (blackspot susceptible) and brown mustard (B. juncea, blackspot susceptible) was investigated. The results of this investigation indicate that destruxin B is transformed to less toxic metabolites by the resistant mustard at a significantly faster rate than that of susceptible plants. These results will be presented and the important implications will be discussed.
KEYWORD: Alternaria brassicae; destruxin B; detoxification; Brassica napus; B. juncea; Sinapis alba
INTRODUCTION
Alternaria blackspot, one of the most destructive fungal diseases of rapeseed (Brassica napus and B. rapa) and brown mustard (B. juncea) (Saharan, G. S., 1993), is caused by the fungal pathogen Alternaria brassicae (Berk.) Sacc. This pathogen infects the aerial plant parts, causing chlorotic and necrotic foliar lesions (Verma, P. R. and Saharan, G. S., 1994). Fruit infections result in premature shattering leading to seed losses during harvesting (Bains and Tewari, 1987). The fungus produces large numbers of spores which may be spread by wind and splashing rain. Dissemination may also occur through wind-blown infected plant tissue, although the main means of spread to new fields is through use of infested seed.
A. brassicae produces a complex mixture of phytotoxins, some of which are host-selective, i.e. toxic only to plants that host the pathogen. The depsipeptide destruxin B is the major host-selective phytotoxin produced both in vitro (Ayer, A. W. and Peña-Rodriguez, L. M, 1987; Bains, P. S. and Tewari, J. P., 1987) and in planta (Buchwaldt, L. and Jensen, J. S., 1991; Pedras and Smith, 1997) by A. brassicae. Physiological effects of destruxin B on diverse Brassica species and other crucifers have been reported (Bains, P. S. and Tewari, J. P., 1987; Buchwaldt, L. and Green, H., 1992; Shivanna, K. R. and Sawhney, 1993), yet the chemistry involved in the plant-pathogen interaction is not understood.
As part of a research program aimed at understanding mechanisms of plant disease resistance (Pedras, 1998), we are evaluating the role of host-selective phytotoxins. Because white mustard (Sinapis alba) is resistant to Alternaria blackspot, we were interested in examining its response to the phytotoxins produced by the pathogen. White mustard is a cruciferous plant grown as a source of condiment and industrial oil in several parts of the world (Hemingway, 1995). To determine a possible correlation between Alternaria blackspot resistance and phytotoxin metabolism, the biotransformation of destruxin B (figure 1) by resistant (white mustard - Sinapis alba) and susceptible (brown mustard - Brassica juncea and rapeseed - Brassica napus) cruciferous tissues was studied. The results of these experiments are reported and implications are discussed.
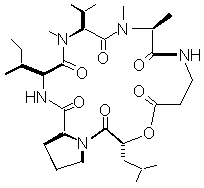
Figure 1. Chemical structure of the phytotoxin destruxin B
METHODOLOGY
Radiolabeled destruxin B was synthesized as previously reported for unlabeled destruxin B (Ward et al. 1997), but utilizing 14C-labeled b-alanine. HPLC (high pressure liquid chromatography, instrument equipped with both ultraviolet and radioisotope detectors) analysis of plant extracts was conducted as previously reported (Pedras and Smith, 1997).
Time-course experiments were conducted by administering radiolabeled destruxin B to petiolated leaves of Brassica napus (susceptible), B. juncea (susceptible), and Sinapis alba (resistant) and incubating leaves in a growth chamber for different periods. The leaf tissue was frozen in liquid N2 and extracted with ethyl acetate, followed by methanol. The extracts were filtered, dried over Na2SO4 and concentrated under reduced pressure. The leaf residues were analyzed by LSC (liquid scintillation counting, ca. 80-90% of recovered radioactive material), and by HPLC. Analysis of “hot” leaf extracts indicated that destruxin B was metabolized by white mustard within 48 hours to a polar metabolite A, which was further metabolized to a more polar metabolite B within 4 days.
Scale-up experiments with cold destruxin B allowed isolation of both metabolites A and B; analysis of spectroscopic data (high resolution electron impact mass spectrometry, proton and carbon nuclear magnetic resonance, and infrared spectroscopy) allowed the elucidation of the chemical structure of metabolite A.
RESULTS AND DISCUSSION
Chemical structure elucidation of metabolite A showed that the transformation of destruxin B by the resistant plant occurred through an oxidation of the g-carbon of the hydroxyacid residue, leading to hydroxydestruxin B. Following the structure elucidation of hydroxydestruxin B, its biotransformation by resistant and susceptible brassica tissues was examined. The HPLC chromatograms (radiodetection) of the extracts showed that the transformation of hydroxydestruxin B by blackspot susceptible rapeseed into a more polar metabolite B occurred at a faster rate than in brown mustard or white mustard (figure 2).
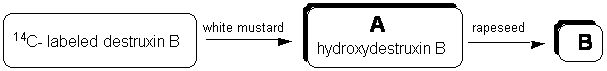
Figure 2. Metabolism of destruxin B by white mustard and rapeseed.
Most significantly, bioassay results utilizing whole plants and cell cultures indicated that hydroxydestruxin B was less toxic than destruxin B to white mustard, rapeseed, and brown mustard, and that the hydroxydestruxin B metabolite (metabolite B) had no detectable phytotoxicity on any of the three species tested. Therefore, our work established that the transformation of destruxin B to hydroxydestruxin B by white mustard represents the rate determining metabolic step in the detoxification pathway. Because hydroxydestruxin B can be metabolized efficiently by rapeseed, it appears that formation of hydroxydestruxin B is the crucial detoxification step.
CONCLUSION
The recent results obtained in our Laboratory have important practical applications: the newly discovered biochemical trait of white mustard, i.e. destruxin B detoxification, could guide the discovery of specific disease resistance enzyme(s) / gene(s). If the blackspot disease resistance of white mustard is related with the presence of specific detoxifying enzyme(s), this trait can be transferred to susceptible brassicas and may improve their resistance to this destructive fungal disease.
ACKNOWLEDGMENTS
We gratefully acknowledge the financial support of the Natural Sciences and Engineering Research Council of Canada (strategic grant to M.S.C.P. and D. E. W.) and the University of Saskatchewan.
REFERENCES
Ayer, A. W.; Peña-Rodriguez, L. M. J. Nat. Products, 1987, 50, 400-407.
Bains, P. S.; Tewari, J. P., Physiol. Mol. Plant Pathol., 1987, 30, 259-271.
Buchwaldt, L.; Green, H. Plant Pathol ., 1992, 41, 55-63.
Buchwaldt, L.; Jensen, J. S. Phytochemistry, 1991, 30, 2311-2316.
Hemingway, J. S. In Brassica Oilseeds, Production and Utilization; Kimber, D. and McGregor, D. I., edis.) CAB International, Wallingford, 1995, pp 373-383.
Pedras, M.S.C. Recent Research Developments in Agricultural and Food Chemistry, 1998, 2, 513-532.
Pedras, M.S.C.; Smith, K.C. Phytochemistry, 1997, 46, 833-837.
Saharan, G. S. in “Breeding Oilseed Brassicas”; Labana, K. S.; Banga, S. K., Editors: Springer-Verlag: Berlin, 1993, pp 181-205.
Shivanna, K. R.; Sawhney, V. K. Theor. Appl. Genet. 1993, 86, 339-344.
Verma, P. R.; Saharan, G. S. in “Monograph on Alternaria Diseases of Crucifers”; Research Branch, Agriculture and Agri-Food Canada, Saskatoon Research Center, Saskatoon, 1994.
Ward, D.E.; Lazny, R.; Pedras, M. S. C. Tetrahedron Lett., 1997, 38, 339-342.