Host Plant acceptance by the pollen beetle (Meligethes aeneus) of a wild Brassicaceae species: Barbarea verna
M. Sc. Desirée Börjesdotter
Swedish University of Agricultural Sciences
Department of Ecology and Crop Production Science
P. O. Box 7043, SE-750 07 Uppsala, SWEDEN
desiree.borjesdotter@evp.slu.se
Abstract
New crops that do not cross or recombine with Brassica species are needed for development of new oil qualities to supply the demand of alternatives to fossil oil products in our society. The species Barbarea verna (Mill.) Asch. is being domesticated to meet this demand. B. verna is biennial and adapted to the climate of northern Europe. Selection for genotypes with advantageous characteristics is a long-term process, 10-15 years being unexceptional. While agronomic properties are being improved, it is also important to study and select for resistance to pests.
This study was initiated to determine whether the pollen beetle causes damage to the species B. verna and whether nitrogen fertilisation interacts with the response of the species B. verna to the potential pest in a field situation.
Two similar split-plot experiments were established with four levels of nitrogen as the main-plot factor (3, 7, 11 and 15 gm-2) and three different pollen beetle treatments as the sub-plot factor. The experiments included three replicates and were carried out at two locations, Kristianstad and Svalöv.
No significant effects of the pesticide treatments were recorded on either dry weight of seed (g seed DW m-2) or any other variable observed. The number of lost pods due to damage to buds by the pollen beetle was low in all experimental treatments. Consequently, no interactions between pesticide treatment and nitrogen were found. However, there were significant differences in seed yield between the experiments and between the nitrogen levels within each experiment.
It seems that the damage by the pollen beetle was limited to a level tolerated by the plant species. The lower reproduction capacity of the pollen beetle on this plant species is a great advantage and certainly increases its potential for cultivation.
Keywords potential oil crop, nitrogen, pesticide treatments
Introduction
The pollen beetle, the species Meligethes aeneus Fab. (Coleoptera: Nitidulidae) in particular, is a pest of great economic importance, attacking oil seed brassicas (Brassica napus var. oleifera L and Brassica rapa var. oleifera L) in Sweden. There is strong motivation for the development of alternative strategies to decrease the use of pesticides in the cultivation of oil crops (Nilsson, 1987). The pollen beetle feeds on pollen from a large number of plant families, but has specialised in only ovipositing in buds of the plant family Brassicaceae. Damage is done by adults and larvae feeding on buds and flowers of the plants (Fritzsche, 1957; Williams and Free, 1978). The extent to which the pollen beetle females accept different brassica species as oviposition sites varies more than their adult feeding behaviour. The species Barbarea verna (Mill.) Asch. is being domesticated to meet the demand for new oil qualities. B. verna is biennial and adapted to the climate of northern Europe. Weak seed dormancy and homogeneous maturation of the plant are other advantages of this species. Since there is a natural variation among individuals, the problems with weak shedding resistance and small seed could be solved (Merker and Nilsson, 1995). Selection for genotypes with advantageous characteristics is a long-term process, which can take 10-15 years or more. While agronomic properties are being improved, it is also important to study and select for resistance to pests. In a no‑choice greenhouse experiment a majority, 74%, of the pollen beetle females never oviposited on the species B. verna (Börjesdotter, unpublished data). On average, each of the egg-laying females laid 1.5 eggs per 5-day period. Under similar conditions in a greenhouse experiment Ekbom and Borg (1996) found that every pollen beetle female laid an average of 4.2 eggs per 2-day period on four different Brassica species (Brassica carinata Braun, B. juncea L Czern., B. napus and B. rapa).
This study was initiated to determine whether the pollen beetle causes damage to the species B. verna and if nitrogen fertilisation interacts with the response of the species B. verna to the potential pest in a field situation. We need to learn more about host plant acceptance by the pollen beetle to be able to develop new oil crops and to improve the brassica oil crops of today.
Material and method
 |
Two similar split-plot experiments were established with four levels of nitrogen as the main-plot factor and three different pollen beetle treatments the sub-plot factor. The experiments included three replicates and were carried out at two locations, one in Svalöv (55°25'N, 13°07'E) on a loam containing 3-6% organic matter and one in Kristianstad (56°10'N, 14°04'E) on a sandy soil containing 6‑12% organic matter. Details of performance and development are listed in Table 1. Due to inappropriate weather conditions in the spring the experiment in Svalöv was established at the beginning of June 1997 and sown in a pure stand. The same seed rate was used in both experiments. The plots in Svalöv were 3.5x8 m and in Kristianstad they were 4x8 m. In August 1997 the barley was harvested and the straw was removed. Plant status and plant density were recorded both in October 1997 and in March 1998. In both experiments the development of the plant stand was recorded according to the scale of Harper and Berkenkamp (1975). In both experiments soil was sampled before fertiliser was applied in the spring of 1998 and after harvest. In each experiment six places were randomly chosen and six cores were taken from 0-60 cm at each site. The soil samples were immediately deep-frozen and not thawed until analysis. The samples were extracted with 2 M KCl. The concentrations of NO3-N were determined using the colorimetric cadmium reduction method (Grasshoff, 1964; Wagner, 1974). The concentration of NH4-N was determined with a spectrometric method by which a coloured complex of NH4-N and sodiumsalicylate is formed in the presence of prussidic acid and chloride (Benesch and Mangelsdorf, 1972). All analyses were made with the AutoAnalyzer TRAACS 800. The analytical values were transformed to gm-2 using average bulk densities and actual gravimetric water contents.
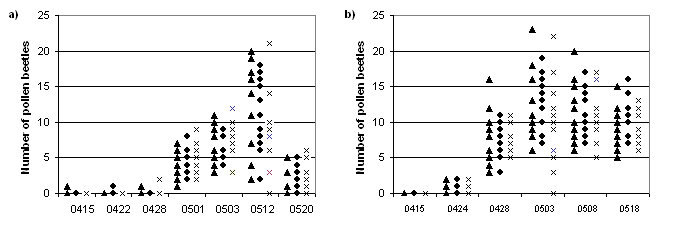
In 1998, the first pollen beetle was spotted in the experimental fields on April 15. The number of pollen beetles in the experimental plots was counted seven times at Kristianstad and six times at Svalöv (Figure 1). In each plot, the pollen beetles were counted on five plants with 10 plant intervals in one row of each plot. On every occasion the starting point was random. At first flowering stage two traps, shallow bowls with water and soap, were placed in each treatment to collect larvae of the first larval instar leaving the plants to pupate in the soil. The traps were changed twice, with one week in between and the contents were analysed.
At Kristianstad, pesticide, Sumi-alpha 0.02 ml m-2 (a. i. Esfenvalerat), was applied once at the second bud stage and once at the third bud stage. At Svalöv, one treatment was to apply pesticide, Decis 0.03 ml m-2 (a. i. Deltametrin), at the third bud stage and another to apply pesticide, Decis (0.03 ml m-2) twice, at the third bud stage and the first flowering stage. In both experiments one treatment was the control.
The flowering time, flowering duration and maturation of the plant stands were recorded. The height of the plants was measured at the second maturation stage (Table 1).
Two weeks before harvest, plant samples were analysed from within an area of 0.25 m-2 in each treatment. Number of plants, number of pods and number of aborted pods were counted and the dry weight of plants was measured for this area.
In order to avoid difficulties caused by the weed Matricaria indora L. at harvest, the experiment in Kristianstad was desiccated with Reglone 0.15 ml m-2 (a. i. Dikvat) four days before estimated harvest date. The shedding of seeds in each plot was estimated the day before harvest through random sampling. The selected plants were visually graded and left in the plots. The number of pods with signs of shedding was counted on nine plants in each treatment. Both experiments were harvested with a combine. Only the central third of the plot was harvested to minimise edge effects. The seed was dried and rinsed before the seed yield was weighed out. Seed samples were dried for 24 h at 105 °C to give a measure of their dry weight.
The data were analysed in a mixed model of
SAS (Littell et al., 1996). The curve fittings in Figure 3 are least
square fittings, which are shown only to guide the eye.
Figure 1. Number of pollen beetles on five plants in twelve plots of each treatment a) on seven occasions at Kristianstad (tcontrol, h pesticide 0422, r pesticide 0428) and b) on six occasions at Svalöv (tcontrol, h pesticide 0428, r pesticide 0428+0503).
Results
Neither of the pesticide treatments affected the seed yield significantly in any of the experiments (Table 2). Consequently, no interaction was found between pesticide treatments and nitrogen fertilisation. However, there were significant differences in seed yield between the experiments and between the nitrogen levels within each experiment (Tables 3 & 4). In general, both the nitrogen utilised and the seed yield of the experiment at Kristianstad at a certain nitrogen level were higher than those of the Svalöv experiment (Figure 2).
 |
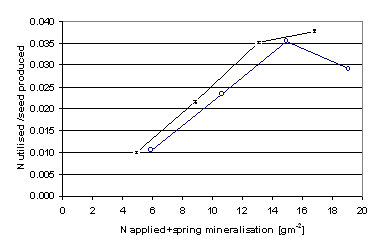 |
Figure 2. The amount of nitrogen utilised per unit DW of seed produced over the total amount of applied and mineralised nitrogen in the spring in Kristianstad (—V—) and in Svalöv (—o—).
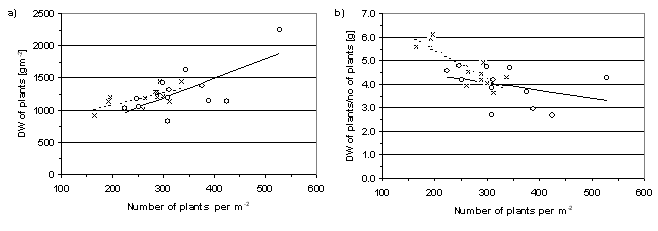
Both experiments were successfully established. The difference in sowing date between the two experiments seemed to have levelled out before the winter. The winter was not severe but for long periods of time the plants were not covered by snow while the temperature was low. In both experiments the plant density had decreased on average by 5% during the winter. However, the plant density was higher and the range was larger at Svalöv than at Kristianstad. Generally, in both experiments the above-ground biomass increased with increasing plant density. Furthermore, the weight of each plant decreased with increasing plant density (Figure 3). The number of pods per dry weight of plant was similar in both experiments and not affected significantly by any of the treatments.
Figure 3. a) Total amount of plant DW and b) The amount of DW per plant in two experiments at Kristianstad (r , - - - -) and at Svalöv (o , ——).
 |
In neither of the experiments did the number of pollen beetles exceed the threshold number of summer rape (½ pollen beetle per plant) for pesticide treatment at the first and second bud stages. In Svalöv it was exceeded at the third bud stage but in Kristianstad it was not exceeded until the first flowering stage. In the later stages of the season, this number was well exceeded (Figure 1). The feeding by adult pollen beetles continued through the flowering stages. Only adult pollen beetles (no larvae) were caught in the traps.
In both experiments the number of aborted pods tended to be lower in plots with pesticide treatment (1.3%) than in the plots without (3.8%).
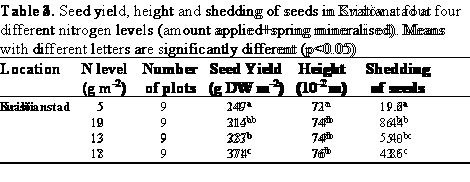 |
There were no differences between the pesticide treatments with regard to plant growth and development. However, nitrogen level had a small influence on seed development. This difference in maturation was obvious since all plant stands within nitrogen treatments matured uniformly. The shedding of seeds was hard to estimate without disturbing the plants. In both experiments, the shedding of seeds was larger at the lower nitrogen level than at the higher. Furthermore, the plants were significantly shorter at the lowest nitrogen level than in the rest of the nitrogen treatments in both experiments (Tables 3 and 4).
Discussion
There was no effect of the pesticide treatments in the experiments. Consequently, the damage on B. verna by the pollen beetle did not affect the seed yield. A low pollen beetle pressure might cause the lack of effect in the first and second bud stages. However, the number of pollen beetles in the experiment at Svalöv well exceeded the threshold number before and after the experimental plots were treated with pesticide. Another reason might be that B. verna is not accepted by, or suitable as a host plant for the pollen beetle.
Preferentially, but not exclusively, the pollen beetle females lay their eggs in buds of 2-3 mm in length (Fritzsche, 1957; Nilsson, 1988; Ekbom and Borg, 1996). Brassica oilseed appears to be more sensitive to attack by the pollen beetle during the medium bud stage (Nilsson, 1987). The thousand seed weight of this specific accession of B. verna was on average 1.19 g. The seed and the seed-producing organs have not been enlarged by plant breeding. However, buds of the preferred size are present on plants of this species (Börjesdotter, unpublished). The presence of different glucosinolate profiles might explain why the female shows preferences for particular plant species, both at oviposition and when feeding. However, it is not yet specified which characteristic, morphological or chemical, is the determining factor for oviposition and feeding for the pollen beetle. In a greenhouse experiment (Börjesdotter, unpublished) the pollen beetle oviposited in buds of B. verna. However the acceptance of this species by the pollen beetle was low. The eggs were laid in the buds of 2-3 mm in length, which are significantly older than buds of that size on, for instance, B. napus. In a field situation it is likely that any eggs laid would not have enough time to hatch and to develop a larva before the bud burst into a flower. In addition, the damage done to the buds of this size subjected to oviposition has, according to Nilsson (1987), little impact on their ability to develop into normal pods. One exception arises when so many eggs are laid in the bud that the larvae produced destroy all the anthers. In such cases the bud will abort. However, a decreased seed yield of plants damaged by the pollen beetle is not merely a response to the loss of buds. A forced change in growth pattern with an increased number of side-branches, severe damage to buds and stems by pollen beetle larvae and environmental factors all contribute to the final seed yield. If the species B. verna avoids damage by the pollen beetle the yield loss due to reorganisation of resources within the plant will not occur. This interaction between the species B. verna and the pollen beetle might be one reason for accepting this potential oil crop. The seeds should not be routinely enlarged during the domestication process without the aspect of pest resistance being taken into consideration.
The response to different nitrogen levels was dependent on the location of the experiment (p<0.05). The estimation of the nitrogen levels available to the plants in the soil was very approximate. However, the response of the plant material verified the accuracy of this simple method for the purpose of these experiments. At Kristianstad, the plants utilised more of the available nitrogen per unit of dry weight of seed produced than at Svalöv (Figure 2). Since the seed yield was significantly higher (p<0.05) at Kristianstad, the total nitrogen uptake was higher than at Svalöv. The actual nitrogen level in the high yielding plots at Svalöv, in terms of seed yield, were probably higher than the average nitrogen level which forms the basis for the ratio in Figure 2. The heterogeneity of nitrogen mineralisation within the blocks is probably a response of the history of this experimental site. The difference in mineralised nitrogen is a positive explanation for why the curve drops at higher amounts of available nitrogen (Figure 2). More advantageous environmental conditions at Kristianstad than at Svalöv contributed to a more effective formation of supporting biomass. At Kristianstad, fewer plants per area utilised more nitrogen and produced a higher seed yield.
The pesticide treatments were applied after the most sensitive developmental stages of the plant material and differed in time and method between the two experiments. The reason for this delay in time was the low pressure of pollen beetles, especially at Svalöv, in the bud stages. The two pesticides used are considered to be equally effective.
Host plant acceptance by the pollen beetle and its relationship to B. verna is a complex subject. However, one conclusion of this work is that the pollen beetle does not damage the B. verna more than the plant material can easily compensate for. A lower reproductive capacity of the pollen beetle on this plant species is a great advantage and certainly increases its potential for cultivation. It is important to continue working to increase the understanding of host plant acceptance by the pollen beetle. Possible resistance characteristics of the B. verna must be identified and preserved in the plant material while agronomic properties of this species are being improved.
acknowledgement
I want to thank Agr. Drs. M. Tuvesson, J. Lundin-Hagman and C. Svensson for helpful comments of my manuscript. The Swedish University of Agricultural Sciences provided financial support for this work.
References
Benesch, R., P. Mangelsdorf, 1972. A method for the colorimetric determination of ammonia in seawater. Helgoländer Wiss Meeresuntersucherung. 23, 365-375.
Ekbom, B., A. Borg, 1996. Pollen beetle (Meligethes aeneus) oviposition and feeding preferences on different host plant species. Entomologia Experimentalis et Applicata 78, 291-299.
Fritzsche, R., 1957. Zur biologie und ökologie der Rapsschädlinge aus der Gattung Meligethes. Zeitschrift für Angewandte Entomologie 40: 222-280.
Grasshoff, K., 1964. Determination of nitrate in sea and drinking water. Kieler Meeresforschung 20, 5-11.
Harper, F. R. & B. Berkenkamp, 1975. Revised growth-stage key for Brassica campestris and B. napus. Canadian Journal of Plant Science 55, 657-658.
Littell, R. C., G. A. Miliken, W. W. Stroup & R. D. Wolfinger, 1996. Common Mixed Models. In: SAS System for Mixed Models, Cary, NC: SAS Institute Inc., pp 31-86.
Merker, A. & P. Nilsson, 1995. Some oil crop properties in wild Barbarea and Lepidium species. Swedish Journal of Agricultural Research 25: 173-178.
Nilsson, C., 1987. Yield losses in summer rape caused by pollen beetles (Meligethes spp). Swedish Journal of Agricultural Research 17, 105-111.
Nilsson, C., 1988. The pollen beetle (Meligethes aeneus F:) in winter and spring rape at Alnarp 1976-1978. II. Oviposition. Växtskyddsnotiser 52, 139-144.
Wagner, R., 1974. A new method for automated nitrate determination in sea water using the AutoAnalyser. Technicon Symposium, Frankfurt am Main, 1974.
Williams, I. H. &J. B. Free, 1978. The feeding and mating behaviour of pollen beetles (Meligethes aeneus Fab.) and seed weevils (Ceutorhynchus assimilis Payk.) on oil-seed rape ( Brassica napus L.). Journal of Agricultural Sciences 91: 453-459.